Consider Volunteering in a Clinical Research Study!
Current: Clinical Trial for Chronic Rhinusitis for Adults with or without Nasal Polyps (TITAN Study)

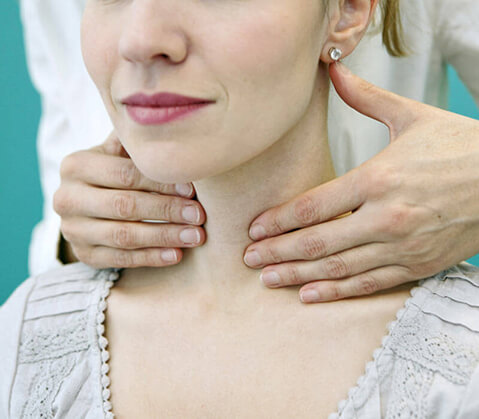
If chronic rhinosinusitis with nasal polyps is causing loss of smell, congestion, runny nose, or facial pain/pressure, you may want to consider a clinical study for adults 18 to 65 years of age. If you qualify, all study-related medication and care are provided at no cost to you.
Why is this study important?
This study is important because it will test how well a study drug works on nasal and sinus symptoms related to chronic rhinosinusitis (persistent swelling of the nasal and sinus passages) for patients with or without nasal polyps (soft growths in the nasal passages). This study will also find out if this study drug is safe.
Even if the study drug does not help you, the results of the study may help others in the future. Pharmaceutical companies use clinical research studies like this one to learn more about investigational medications before they are made available to the public. Study volunteers can help us in this important research. Thank you for considering participation in this trial.
What is the purpose of this study?
The purpose of this research study is to:
- Test the safety of the study drug (called GB001)
- Find out if GB001 helps decrease symptoms of chronic rhinosinusitis such as stuffy nose or loss of smell
- Check how much of the study drug gets into the bloodstream and how long the body takes to get rid of the drug
Who can participate in this study?
To be eligible for this study, you must be:
- 18 to 65 years of age
- Diagnosed with chronic rhinosinusitis with or without bilateral nasal polyps
- Able to give informed consent
This is not a complete list of study requirements. The study doctor will review the full criteria for this study with you.
How long will the study last?
Your participation in this study will last about 24 weeks and will include around 8 study visits to the study center.
What are my costs to take part in this study?
You do not have to pay for the study drug, study supplies, study visits, or tests that are part of the research study.
What risks are involved if I decide to participate?
There are possible risks involved in any clinical study. Your study doctor will review the risks with you, and you will be closely monitored throughout the study. Since the study drug GB001 is investigational, there may be risks that are unknown. Tell your doctor if you have any symptoms. There also may be unknown risks to a pregnancy, embryo, or fetus (unborn baby) if you become pregnant.
If you are interested, please contact Ninos Joseph at (773) 289-1823.