Consider Volunteering in a Clinical Research Study!
Current: Clinical Trial for Treatment of Moderate to Severe Atopic Dermatitis (ZEST Study)
About this study
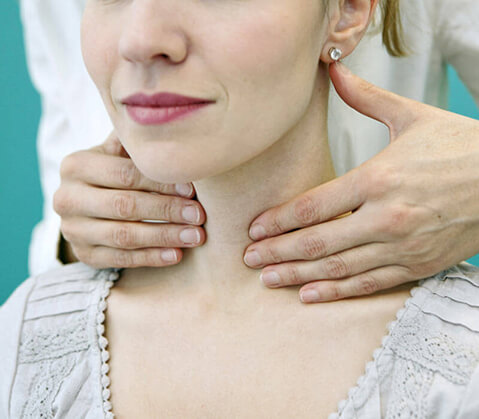
The ZEST trial is a randomized, double-blind, placebo-controlled multicenter dose-ranging study to assess the safety and efficacy of multiple oral ZPL389 doses in patients with moderate to severe atopic dermatitis.
Are you eligible to participate in this study?
You may be eligible for this study if you meet the following criteria:
- Conditions: Dermatitis | Atopic Dermatitis | Atopic
- Age: Between 18 – 100 Years
- Gender: Male or Female
Inclusion Criteria:
- Subjects must give a written, signed and dated informed consent
- Chronic atopic dermatitis present for at least 1 year before Baseline
- Moderate to severe atopic dermatitis defined as per EASI, IGA, and BSA.
- Documented recent history (within 6 months before the screening visit) of inadequate response to treatment with topical medications or for whom topical treatments are otherwise medically inadvisable
- Candidate for systemic treatment
Exclusion Criteria:
- Any skin disease that would confound the diagnosis or evaluation of atopic dermatitis disease activity
- Use of other investigational drugs within 5 half-lives of enrollment, or within 30 days or until the expected pharmacodynamic effect has returned to baseline, whichever is longer.
- History of hypersensitivity to any of the study drug constituents or to drugs of similar chemical classes.
- Participation in prior ZPL389 studies
If you are interested, please contact Ninos Joseph at (773) 289-1823.